Latest News
Latest News
THE OPHTHALMOLOGY MEDICINES COMPANY
Kodiak Sciences is a biopharmaceutical company committed to researching, developing and commercializing transformative therapeutics to treat high prevalence retinal diseases
OUR MISSION
Kodiak aims to prevent and treat the leading causes of blindness in the developed world

TRAILBLAZING SCIENCE
OUR CREATIVE AND THOUGHTFUL FOUNDATION

“GO-TO” MEDICINES
OUR CHALLENGE TO THE STATUS QUO

SINGULAR FOCUS IN OPHTHALMOLOGY
OUR 24/7/365
OUR PRODUCT CANDIDATES
We intend to progress our portfolio of three late-stage clinical assets as rapidly as we can into Phase 3 value inflection points
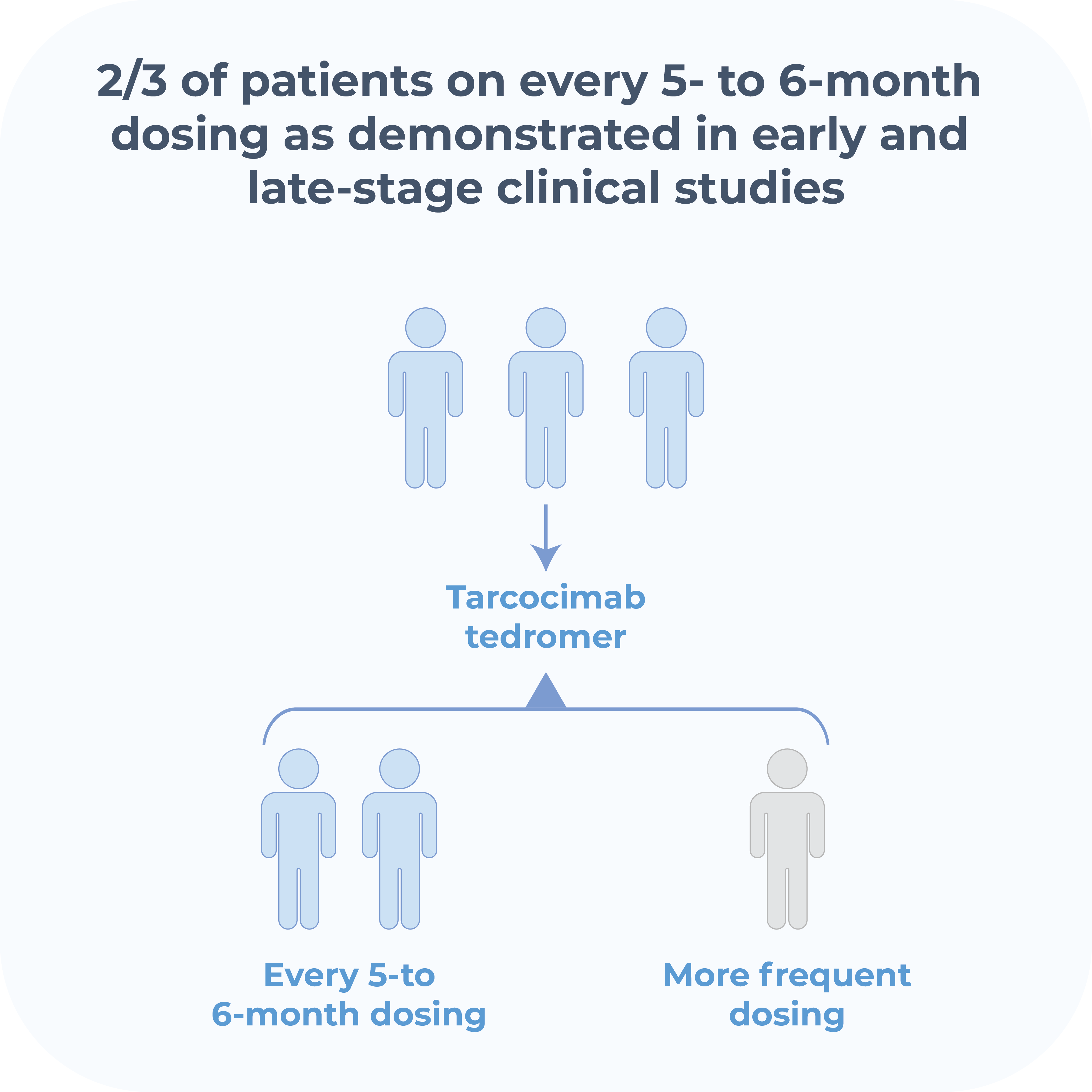
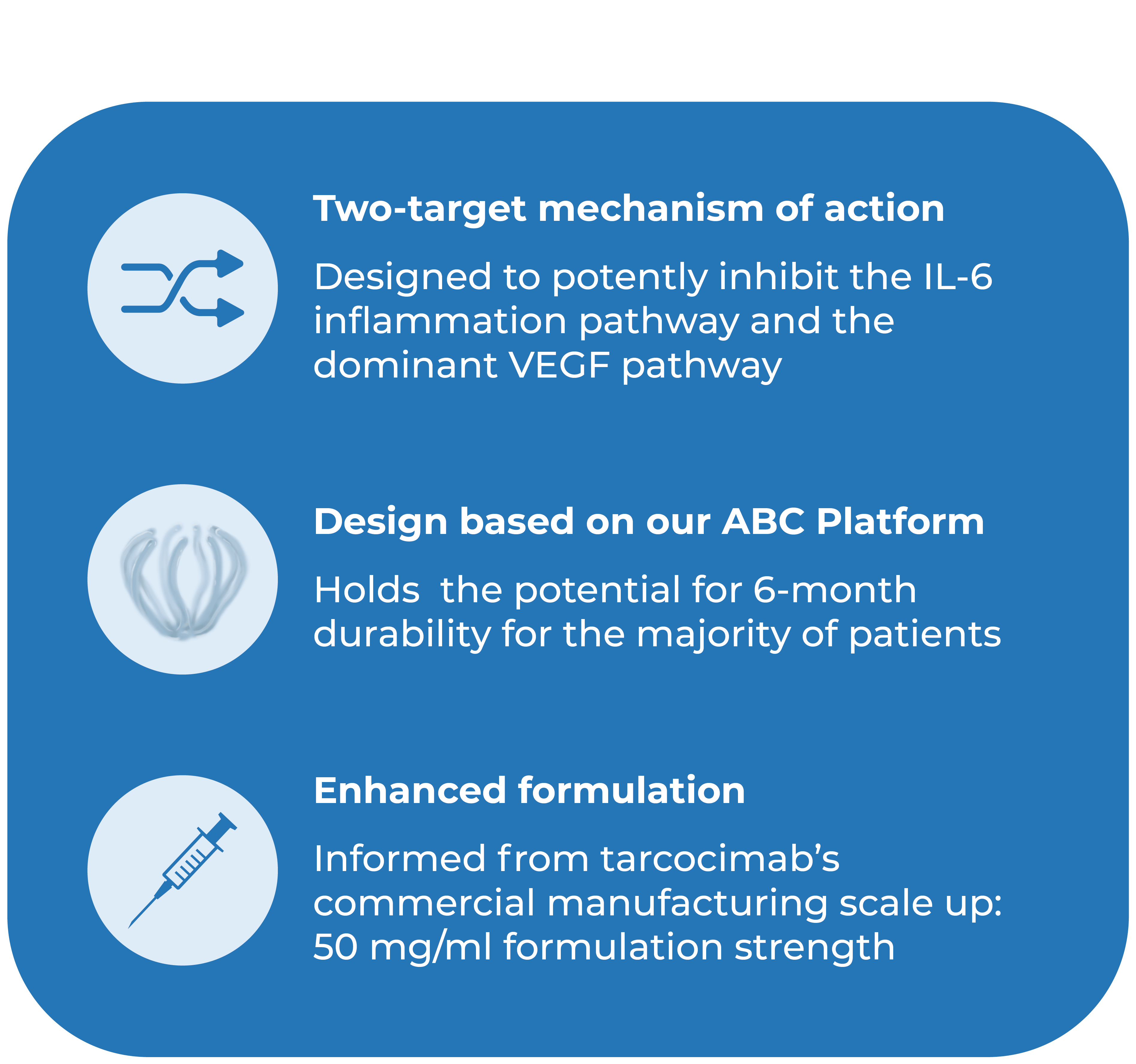
Tarcocimab Tedromer
High-prevalence retinal
vascular diseases
Diabetic retinopathy
Retinal vein occlusion
Wet AMD
Phase 3
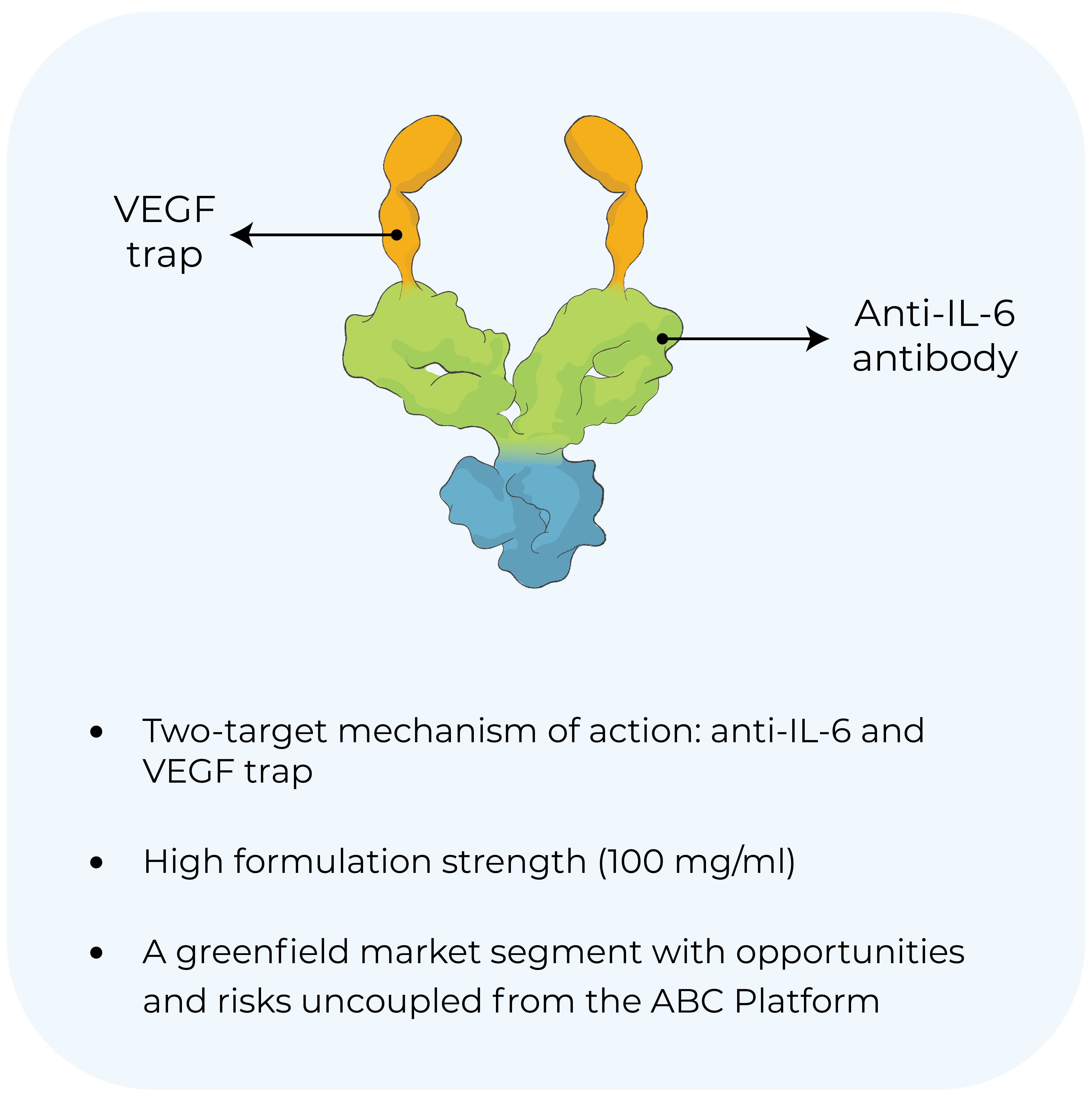
KSI-501
High-prevalence retinal
vascular diseases
Wet AMD
Diabetic macular edema
Phase 3 study initiation targeted mid-2024
KSI-101
Macular edema
associated with inflammation
Phase 2b/3 study initiation targeted 2024
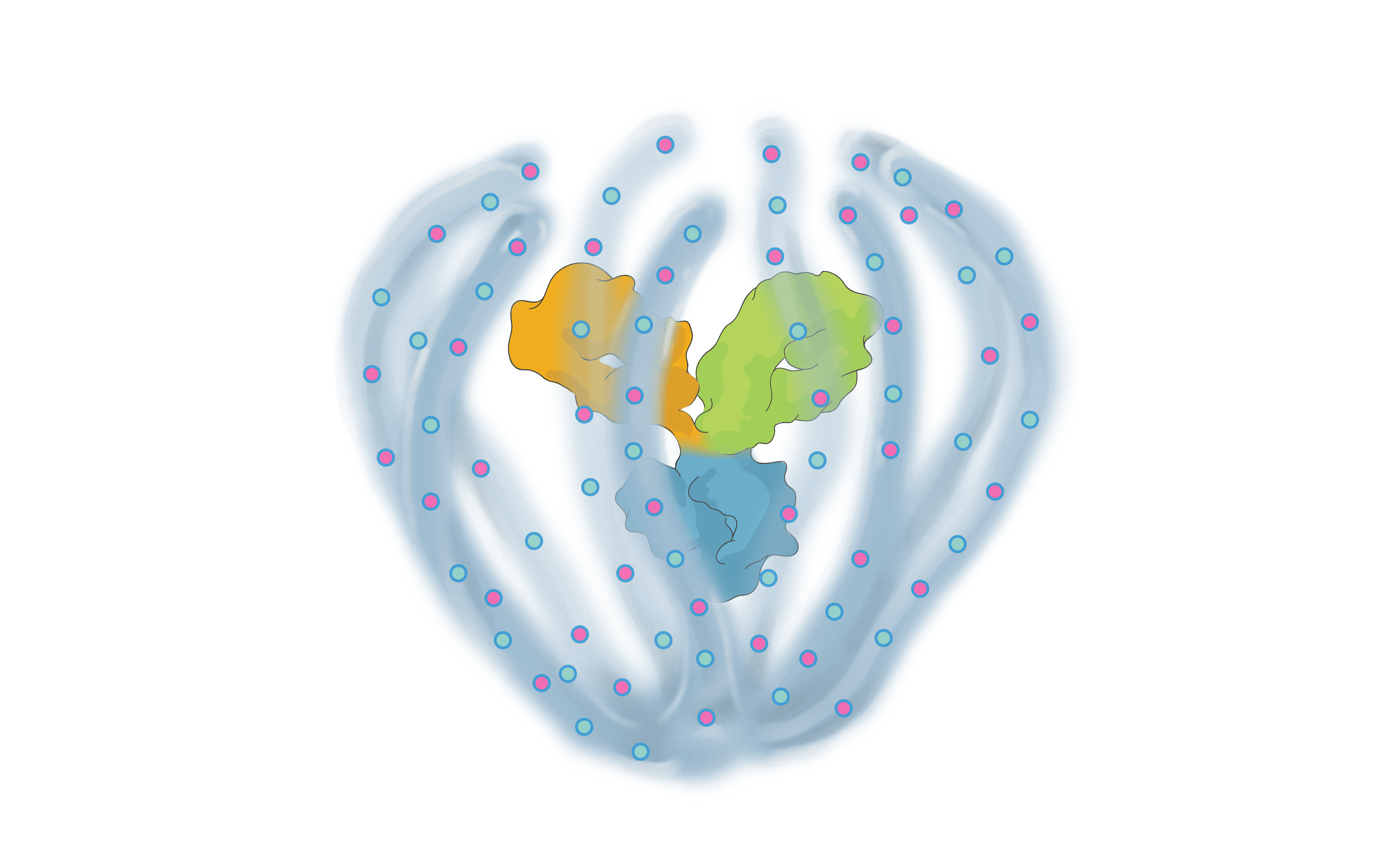
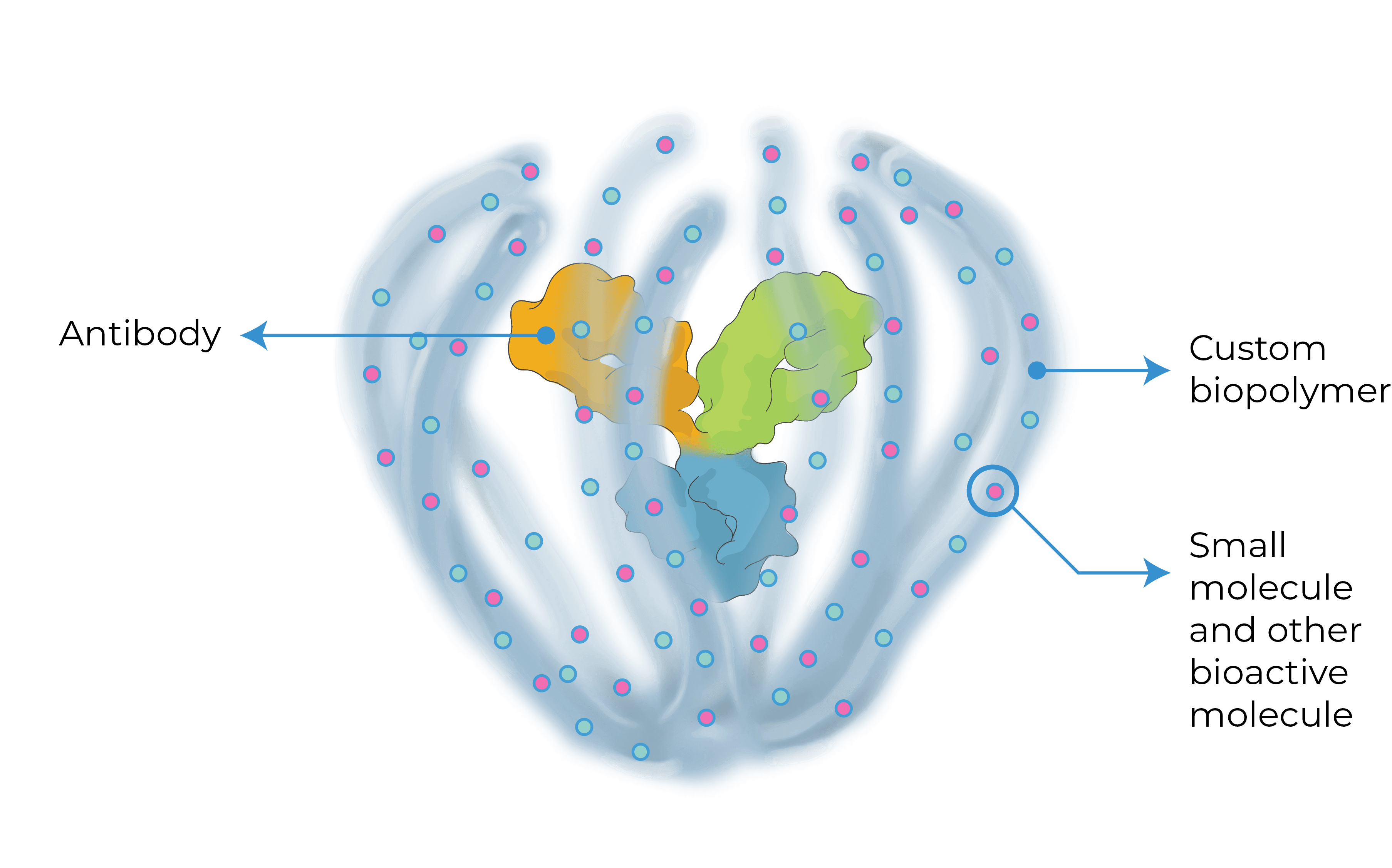
THE ABC OF OUR SCIENCE
We are bringing new science to the design and development of next generation retinal medicines. Our Antibody Biopolymer Conjugate (ABC) Platform uses a bio-inspired polymer to enable multi-modal, multi-mechanistic treatment of complex ocular and systemic diseases
Antibody Biopolymer Conjugate
- Designed-in durability
- Designed to target multiple intracellular and extracellular mechanisms simultaneously
- Designed for ocular and systemic diseases
OUR CULTURE AND VALUES
We are actively building a place, our Kodiak Village, where we are inspired, where we feel valued, and where we can grow our personal and professional journeys while advancing our mission. Historically referred to within Kodiak as the 4 C’s, our values are:

CURIOSITY
Staying fresh.
Asking why.
Being naturally inquisitive.

CREATIVITY
A safe place to think outside the box.
A focus on how to solve the problem.

COMPASSION
For ourselves.
For our colleagues.
For our patients.

COURAGE
Saying no when that’s the right answer.
Saying yes when that’s the right answer.
Regardless.
WORKING AT KODIAK
We aspire to global leadership in ophthalmology through internal focus and by aggregating top talent, technologies, discoveries and ideas.